Low-Endotoxin Recovery (LER) in Biopharmaceutical Products
Low-endotoxin recovery (LER) has emerged as a critical analytical and patient-safety concern in the biopharmaceutical industry. Endotoxin testing is a regulatory requirement for parenteral and implantable products, and failure to accurately detect endotoxin can result in the release of products with clinically significant pyrogenic risk [1,2].
Over the past decade, multiple investigations have demonstrated that certain biopharmaceutical formulations can progressively reduce the detectable activity of endotoxin over time, despite the endotoxin remaining biologically active. This phenomenon, termed low-endotoxin recovery, challenges traditional assumptions underlying the bacterial endotoxins test (BET) and needs enhanced understanding, risk assessment, and control strategies [3,4].
What Is Low-Endotoxin Recovery?
Low-endotoxin recovery is defined as a progressive masking or inactivation of detectable endotoxin activity in a sample, resulting in recovery of a known endotoxin spike at less than 50% of its expected value [4,5]. While there is ongoing debate regarding the distinction between classical assay interference and LER, a key differentiating feature is the time dependency of the effect. Conventional assay interference is typically immediate and constant, and can often be overcome by simply diluting the sample, whereas LER develops gradually over time until reaching a stable equilibrium. This temporal characteristic is central to both identification and mechanistic understanding of LER [3,6].
Importantly, LER does not necessarily imply destruction or removal of endotoxin. Instead, it reflects a reduction in measurable endotoxin activity relative to the reference standard, typically control standard endotoxin, as detected by endotoxin assays [4,7].
Why Low-Endotoxin Recovery Happens?
Endotoxin, or lipopolysaccharide (LPS), is an amphipathic molecule consisting of a hydrophobic lipid A moiety and a hydrophilic polysaccharide region. This dual nature confers surfactant-like properties, enabling endotoxin molecules to self-associate into homo-supramolecular structures such as micelles, vesicles, and aggregates. The structural organization of these assemblies is highly sensitive to environmental conditions, including pH, temperature, ionic strength, and the presence of divalent cations (such as calcium and magnesium) [7-9].
The affinity of endotoxin for the components of the drug or drug matrix can also lead to LER. The phenomenon was first recognized in protein-based formulations, where spiked endotoxin exhibited strong binding to therapeutic proteins, resulting in recoveries below 50% [4]. Subsequent studies have demonstrated that LER can occur across a wide range of biopharmaceutical modalities, including monoclonal antibodies, vaccines, cell and gene therapies, lipid-based formulations, and nucleic acid products [3,5].
Matrix components implicated in endotoxin masking include proteins, surfactants, chelating agents, lipids, and excipients, which are commonly used for stabilisation [3,5-6,9].
Crucially, the biological potency of endotoxin is closely linked to its aggregation state. Since all regulatory approved endotoxin assays measure biological activity rather than mass concentration, changes in endotoxin conformation or aggregation can lead to reduced assay responsiveness without there being a concurrent reduction in the actual endotoxin burden [7].
Are all products and samples likely to demonstrate LER?
Not all samples are susceptible to LER. Water samples — including water for injections, purified water, dialysis waters — will not demonstrate LER. The same is true for many small molecular weight drug products that are typically associated with relatively simple formulations. However, as the complexity and molecular weight of the sample components increase, the chances of endotoxin-component interactions also increase. There is an increasing awareness that complex macromolecular biopharmaceutical formulations — therapeutic proteins, nucleic acid products and cell therapies — can be prone to LER. To complicate the situation further, each step in a multi-step biomanufacturing process can present a new LER scenario.
Is LER confined to injectable products?
Whilst the main risk lies with complex parenteral biopharmaceutical and cell therapy products, LER can influence endotoxin testing results for other types of products. In 2023, the FDA sent a warning letter to a major manufacturer of collagen-based medical devices used in wound care, soft tissue repair and reconstructive surgery, identifying issues with weak endotoxin testing method validation and concerns that LER was influencing the endotoxin levels reported. This resulted in a wide-ranging product recall [10].
How to Detect Low-Endotoxin Recovery?
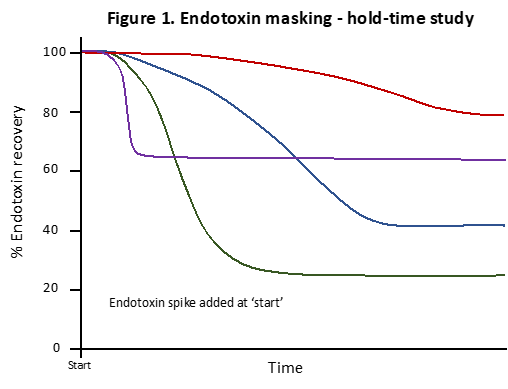
The most reliable method for identifying LER is the performance of endotoxin hold-time studies. In these studies, a known quantity of endotoxin is spiked into the product matrix and tested immediately, followed by repeated testing at predefined intervals over time. A progressive decline in spike recovery to below 50% (figure 1) is indicative of LER [4-5].
Figure 1 shows a range of possible endotoxin masking-time profiles resulting from the addition of an endotoxin spike on day 0. Notice that not only does the maximum degree of masking vary but the time it takes to achieve this maximum level also varies. A range of widely varying profiles can be encountered, and indeed the time axis in figure 1 may span multiple days.
Hold-time studies may need to extend over days or weeks to fully characterize the masking kinetics, as some formulations exhibit slow-onset LER [3]. There is also a demonstrable temperature-dependent nature to LER; higher temperatures generally accelerate masking whilst lower temperatures slow the process down. It is therefore recommended that LER time-hold studies are conducted at both room temperature and at 2 – 8oC. Such studies are increasingly recognised as a critical component of method suitability assessments, particularly for complex biologics. Harmonized protocols developed by industry consortia, such as the BioPhorum Operations Group (BPOG) harmonized LER study protocol and the Parenteral Drug Association Technical Report No. 82 on Low Endotoxin Recovery, have improved consistency in LER evaluation. Regulatory Q&A documents, such as those from the European Medicines Agency, also provide harmonized expectations for LER study design and reporting, although interpretation and product-specific risk assessment remain necessary for each formulation.
Regulatory and Patient Safety Implications
The primary risk associated with LER is the inadvertent release of biopharmaceutical products containing higher endotoxin levels than indicated by routine testing. Since endotoxin may retain its biological activity despite being masked from detection, patients may be exposed to pyrogenic doses capable of inducing fever, inflammation, hypotension, or, in severe cases, septic shock–like reactions [2,8].
Regulatory agencies, including the U.S. Food and Drug Administration, have acknowledged LER as a legitimate concern and have emphasized the need for robust method qualification and risk-based controls [2]. Failure to appropriately address LER can lead to regulatory issues, product recalls, and significant patient harm. Since the discovery of LER in 2013, the FDA has frequently withheld approval for Biologic License Applications (BLAs) if the sponsor cannot provide "hold-time" study data demonstrating that endotoxins remain detectable in the product over time. Consequently, understanding and mitigating LER is not merely an analytical issue but a core element of product quality and patient safety assurance.
What should you do now?
Reporting endotoxin underestimates or false negatives carries major consequences both for patients and drug manufacturers. Therefore, if you are unsure if LER is influencing your data, performing a hold-time study is the only way to put your mind to rest. Validating and performing such a study to the highest level of confidence can be challenging even for Quality Control laboratories. Contracting a UKAS-accredited endotoxin testing services laboratory to perform your LER studies is one way of assuring competence and quality. In the UK, Cotton Mouton Diagnostics is the only endotoxin testing centre that is UKAS (ISO 17025) accredited to perform LER hold-time and method optimisation studies.
References
United States Pharmacopeial Convention. (2024). United States Pharmacopeia - USP–NF 2024: Chapter <85> Bacterial Endotoxins Test. Rockville, MD: USP.
Center for Drug Evaluation and Research (2012). FDA Guidance for Industry: Pyrogen and Endotoxins Testing. Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-pyrogen-and-endotoxins-testing-questions-and-answers
Tsuchiya, M. (2021). Sample Treatments That Solve Low Endotoxin Recovery Issues. PDA Journal of Pharmaceutical Science and Technology, 73(5): 433-442.
Bolden, J.S. et al. (2014). Evidence Against a Bacterial Endotoxin Masking Effect in Biologic Drug Products by Limulus Amebocyte Lysate Detection. PDA Journal of Pharmaceutical Science and Technology, 68(5): 472-477.
BioPhorum Operations Group. (2024). It is time to reconsider the use of naturally occurring endotoxins in endotoxin recovery studies: Part 2 of a BioPhorum harmonized endotoxin recovery study. https://www.biophorum.com/news/it-is-time-to-reconsider-the-use-of-naturally-occurring-endotoxins-in-endotoxin-recovery-studies-part-2-of-a-biophorum-harmonized-endotoxin-recovery-study/
Reich, J., et al. (2019). Low Endotoxin Recovery—Masking of Naturally Occurring Endotoxin. International Journal of Molecular Sciences, 20(4): 838.
Petsch, D. & Anspach, F.B. (2000). Endotoxin removal from protein solutions. Journal of Biotechnology, 76(2–3): 97–119.
Rietschel, E.T., et al. (1994). Bacterial endotoxin: Molecular relationships of structure to activity. FASEB Journal, 8(2): 217–225.
Gorman, A. & Golovanov, A.P. (2022). Lipopolysaccharide Structure and the Phenomenon of Low Endotoxin Recovery. European Journal of Pharmaceutics and Biopharmaceutics, 180: 289–307.
Integra Lifesciences, FDA Warning letter MARCS-CMS 698850 19th Dec 2024