What Does ‘Low Endotoxin’ Really Mean?
Endotoxin control is critical in pharmaceutical workflows from early-stage research through commercial manufacturing. However, the industry frequently uses broad labels such as “low endotoxin”, “endotoxin-tested”, or even “endotoxin-free” across consumables and reagents without a standardised numerical definition. Unlike Water for Injection (WFI), where specific endotoxin limits are clearly defined in Pharmacopoeias [1,2], many other reagents and consumables carry claims that remain loosely interpreted or inconsistently validated. This lack of clarity can affect data quality, comparability, and regulatory confidence.
The Issue of the ‘Low Endotoxin’ Label
The challenge is straightforward: the terms “low endotoxin” or “endotoxin-free” do not have a universally agreed numerical threshold. For some suppliers, “low endotoxin” may mean <0.5 EU/mL; for others, <5 EU/mL. In some product categories, thresholds are reported per mL, per gram, per device, or not quantified at all. As a result, two products with the same marketing claim may differ in endotoxin concentration by an order of magnitude or more.
Below are different product categories where the same “low endotoxin” or “endotoxin-free” claims mask substantial variability.
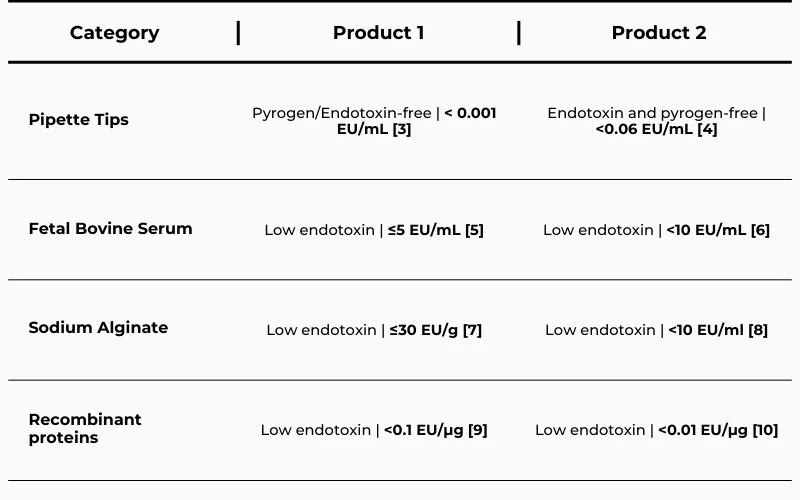
Table 1 – Examples of reagents and consumables used in pharmaceutical research and manufacturing that include the “low endotoxin” or “endotoxin-free” claims while presenting very different endotoxin levels.
These products share similar descriptors — “low endotoxin” or “endotoxin-free” — yet the numerical values tell a different story.
Why Unquantified Endotoxin Labels Matter
The difference between 0.1 EU/mL and 1 EU/mL can have major consequences, especially for biologics, cell therapies, and other endotoxin-sensitive applications where even small variations can shift cell behaviour, immune response, or assay outcomes.
This lack of a universal threshold for the “low endotoxin” and “endotoxin-free” claims can impact research, product development and even the manufacture of biopharmaceuticals by affecting:
Data Reliability: Endotoxin triggers immune and inflammatory pathways. Even small increases can alter cytokine expression profiles, affecting in vitro assay outcomes [11,12].
Reproducibility Across Batches: Variable endotoxin levels can contribute to batch-to-batch inconsistency, a major barrier in biologics research and manufacturing [13].
Comparability Across Vendors: Without standardised reporting, decision-makers cannot directly compare product options on performance risk or regulatory suitability [14].
Compliance & Documentation: Regulatory submissions often require traceable endotoxin values. A label without a quantified level can create ambiguity during audits or process validation [15].
In short, the ambiguity surrounding “low endotoxin” and “endotoxin-free” labels introduces risks that many pharma teams often only realize once variability has already impacted a study or production run.
How CMD Supports Consistency
CMD works with pharma and biotech organisations to ensure that endotoxin testing is clearly defined, validated, and traceable. CMD’s approach emphasizes:
Verified LAL-based endotoxin testing with validated detection ranges
Transparent endotoxin quantification rather than broad marketing labels
Batch-to-batch consistency assurance through controlled quality systems
Support for documentation aligned to regulatory expectations
By grounding endotoxin levels in accurate measurements, not marketing language, CMD helps clients maintain data integrity, comparability, and confidence across research, process development, and regulatory submission phases.
Conclusion
As modern drugs become more biologically complex, endotoxin sensitivity continues to increase, yet the language used to describe endotoxin limits has not kept pace. The terms “low endotoxin” and “endotoxin-free” should be interpreted critically, with attention paid to the actual specification and its suitability for the intended application. Partnering with organisations that precisely quantify endotoxin levels, such as CMD, enables more reliable and reproducible outcomes across the pharmaceutical lifecycle.
References
United States Pharmacopeial Convention (2020). <85> Bacterial Endotoxins Test. In: United States Pharmacopeia and National Formulary (USP–NF). Rockville, MD: United States Pharmacopeial Convention.
European Directorate for the Quality of Medicines & HealthCare - EDQM (2023). 2.6.14 Bacterial Endotoxins. In: European Pharmacopoeia, 11th ed. Strasbourg: Council of Europe.
Pipette Tips – product 1: https://www.eppendorf.com/br-pt/Produtos/Manipula%C3%A7%C3%A3o-de-l%C3%ADquidos/Ponteiras-de-pipeta/epTIPS-p-0030075250
Pipette Tips – product 2. ThermoFisher’s Certificate of Conformity. Lot Number: 21300243. Part Number: T070RNS-FIS. Item Description: 200UL BEV CLR ST 02-707-419960 TIPS / PK, 5 PK / CS
Fetal Bovine Serum – product 1: https://cdn.cytivalifesciences.com/api/public/content/digi-17854-pdf
Fetal Bovine Serum – product 2: https://www.fishersci.at/shop/products/fetal-bovine-serum-fbs-premium-6/30535602
Sodium Alginate – product 1: https://group.nagase.com/nagasechemtex/en/products/product-information/low-endotoxin-arcofeliz/
Sodium Alginate – product 2: https://www.sigmaaldrich.com/specification-sheets/395/021/918652-BULK.pdf
Recombinant Proteins – product 1: https://www.rndsystems.com/products/recombinant-protein-quality
Recombinant Proteins – product 2: https://www.acrobiosystems.com/category/recombinant-proteins/ultra-low-endotoxin-proteins?srsltid=AfmBOop5J3F5gKYLN3JXmLQQlraBiYDrRn-Z7591F-SVMFYD_ZIuT-Sy
Akira, S. & Takeda, K. (2004). Toll-like receptor signalling. Nature Reviews Immunology, 4: 499–511.
Dinarello, C. (2009). Cytokines as mediators of endotoxin effects. Journal of Clinical Investigation, 123(7): 2773–2781.
Schrom, E. et al. (2020). Endotoxin contamination impacts reproducibility in cell culture systems. Frontiers in Cell and Developmental Biology, 8: 559.
US FDA (2012). Pyrogen and Endotoxins Testing: Questions and Answers. Available at https://www.fda.gov/regulatory-information/search-fda-guidance-documents/pyrogen-and-endotoxins-testing-questions-and-answers
European Medicines Agency (EMA) (2013). ICH Q4B Annex 14 Bacterial endotoxins tests - Scientific guideline. Available at https://www.ema.europa.eu/en/ich-q4b-annex-14-bacterial-endotoxins-tests-scientific-guideline